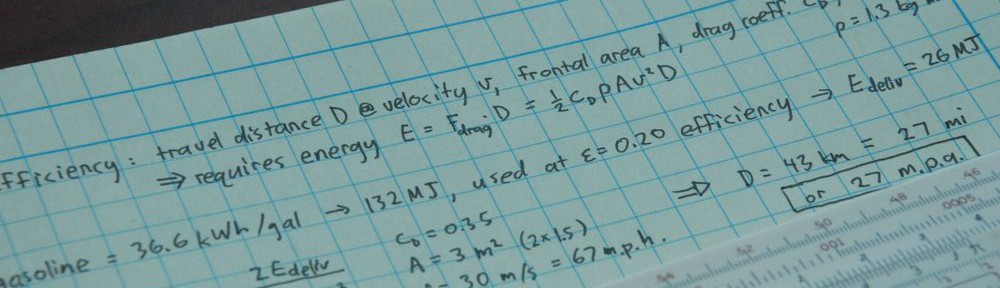[An updated treatment of some of this material appears in Chapter 20 of the Energy and Human Ambitions on a Finite Planet (free) textbook.]
 If you’re one of those humans who actually eats food, like I am, then a non-negligible part of your energy allocation goes into food production. As an approximate rule-of-thumb, each kilocalorie ingested by Americans consumes 10 kilocalories of fossil fuel energy to plant, fertilize, harvest, transport, and prepare. The energy investment can easily exceed a person’s household energy usage—as is the case for me. But much like household energy, we control what we stick in our mouths, and can make energy-conscious choices that result in substantial reductions of energy consumption. I now call myself a flexitarian, a term acknowledging that my body is a flex-fuel vehicle, but also that I need not be rigid about my food choices in order to still make a substantial impact on the energy front.
If you’re one of those humans who actually eats food, like I am, then a non-negligible part of your energy allocation goes into food production. As an approximate rule-of-thumb, each kilocalorie ingested by Americans consumes 10 kilocalories of fossil fuel energy to plant, fertilize, harvest, transport, and prepare. The energy investment can easily exceed a person’s household energy usage—as is the case for me. But much like household energy, we control what we stick in our mouths, and can make energy-conscious choices that result in substantial reductions of energy consumption. I now call myself a flexitarian, a term acknowledging that my body is a flex-fuel vehicle, but also that I need not be rigid about my food choices in order to still make a substantial impact on the energy front.
An earlier post on how many miles per gallon a human gets while walking or biking touched on the fact that fossil fuels undergird our food supply. As a result, walking to the grocery store effectively uses as much fossil fuel as would a typical sedan. The lesson is not to walk less, but to change that 10:1 ratio for the better by eating more smartly. Once upon a time, we put less than one kilocalorie of energy into food production per kilocalorie obtained (or else we and our draft animals would have starved to death). So the 10:1 ratio is not at all inescapable, and depends strongly on the foods we choose to eat.
Continue reading →
Hits: 10326
 The holiday season is upon us, and for many, this translates into a marked uptick in the consumption of tasty food treats. I’m no different, and can really pack it in on such occasions. For instance, the day after Thanksgiving this year, I stepped on the scale to find myself about 5 pounds (~2 kg) above normal weight. I kicked in my diet plan, and by Monday morning (3 days later) I was back to normal. Resume course. I use a simple formula, backed by physics, that works every single time. The topic is Do-the-Math-relevant for two reasons: it applies quantitative physics to everyday life, and it touches on attitudes relevant to energy/resource conservation.
The holiday season is upon us, and for many, this translates into a marked uptick in the consumption of tasty food treats. I’m no different, and can really pack it in on such occasions. For instance, the day after Thanksgiving this year, I stepped on the scale to find myself about 5 pounds (~2 kg) above normal weight. I kicked in my diet plan, and by Monday morning (3 days later) I was back to normal. Resume course. I use a simple formula, backed by physics, that works every single time. The topic is Do-the-Math-relevant for two reasons: it applies quantitative physics to everyday life, and it touches on attitudes relevant to energy/resource conservation.