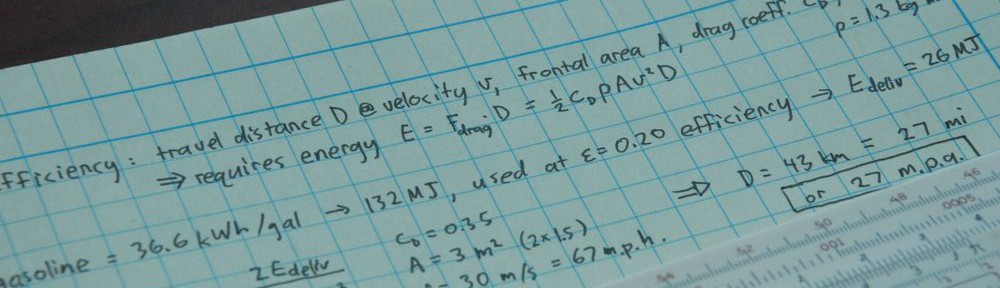The principal challenge of this century, in my view, will be adapting to a life without abundant, cheap fossil fuels. It has been the lifeblood of our society, and turns out to have some really fantastic qualities. The jury is still out as to whether we will develop suitable/affordable replacements. But additional challenges loom in parallel. Water is very likely to be one of them, which is especially pertinent in my region. For true believers in the universality of substitution, let me suggest two things. First, come to terms with the finite compactness of the periodic table. Second, try substituting delicious H2O with H2O2. It has an extra oxygen atom, and we all know that oxygen is a vital requisite for life, so our new product will be super-easy to market. Never-mind the hydrogen peroxide taste, and the death that will surely visit anyone foolish enough to adopt this substitution. Sometimes we’re just stuck without substitutes.
The principal challenge of this century, in my view, will be adapting to a life without abundant, cheap fossil fuels. It has been the lifeblood of our society, and turns out to have some really fantastic qualities. The jury is still out as to whether we will develop suitable/affordable replacements. But additional challenges loom in parallel. Water is very likely to be one of them, which is especially pertinent in my region. For true believers in the universality of substitution, let me suggest two things. First, come to terms with the finite compactness of the periodic table. Second, try substituting delicious H2O with H2O2. It has an extra oxygen atom, and we all know that oxygen is a vital requisite for life, so our new product will be super-easy to market. Never-mind the hydrogen peroxide taste, and the death that will surely visit anyone foolish enough to adopt this substitution. Sometimes we’re just stuck without substitutes.
Substitution silliness aside, water and energy are intimately related in what has been termed the Energy-Water Nexus (see for example the article by Michael Webber from this conference compilation; sorry about the paywall). We’ll explore aspects of this connection here, touching on pumping water, use of water for the production and extraction of energy, and desalination. As glaciers and snowpack melt and drought becomes more common in the face of climate change, our water practices will need to be modified, hitting energy right in the nexus.
Continue reading →
Hits: 4269